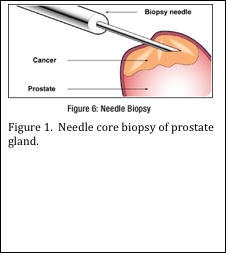
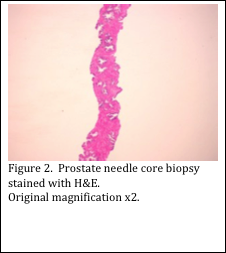
Whether you work in a hospital laboratory or a private reference laboratory, you probably receive specimens of prostate gland. In a hospital setting, the specimen may be received from a transurethral resection of the prostate, or a surgical resection of the entire prostate gland. In both cases, large pieces of prostate tissue are submitted to the pathology laboratory. In a private reference laboratory, usually needle core biopsies of the prostate are received (see Figure 1). These specimens usually measure approximately 1 mm in diameter by 1 cm long (Figure 2). Multiple needle core biopsies are usually procured from different areas within the prostate.
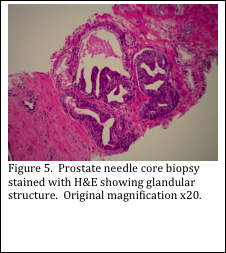
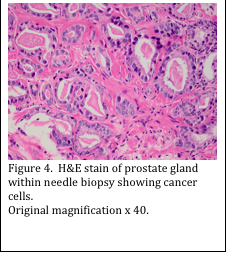
The prostate gland is comprised of many glands, with associated ducts, surrounded by connective tissue (Figure 3). The pathologist must assess the glandular areas and determine if there are any cancer cells present. Sometimes the diagnosis can be made with a hematoxylin and eosin (H&E) stained slide (Figure 4). Other times, the H&E slide may not be conclusive (Figure 5). In this case, immunohistochemical (IHC) stains must be performed.
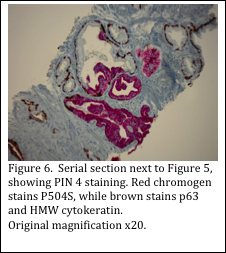
A multi- antibody IHC stain can be used on prostate sections to assist in demonstrating if cancer cells are present. A high molecular weight cytokeratin cocktail of clones CK5 and CK14 are used to stain basal epithelia in the prostate gland. The P504S protein is known to be expressed by prostate adenocarcinoma cells, but not in benign prostate cells. It may also be produced by high grade prostatic intraepithelial neoplasia (PIN). Finally, the p63 antibody is added to the cocktail to detect normal cells, and is not produced by malignant tumor cells. The results of the staining patterns are used to confirm or rule out prostate cancer and prostatic intraepithelial neoplasia (Figure 6).
Initial receipt and handling of prostate needle cores must be done with care. The cores are small, fragile and easily damaged. Many times they are received fragmented. The exact number of cores and fragments must be documented during the surgical grossing procedure. These specimens must be wrapped, or contained in some way to prevent escape from the tissue cassette.
Processing may be done using routine tissue processing through alcohols, xylene and paraffin. A short biopsy protocol should be used to prevent over-dehydration of the tissue. Alternatively, a microwave assisted processor may be used. The omission of xylene during this processing protocol ensures that needle cores remain soft for optimal cutting.
The same care taken in surgical grossing must be utilized during the embedding procedure. Tissues must be unwrapped and handled gently to prevent fragmentation of the cores. Also, the cores must be embedded completely flat, to ensure accurate representation of the tissue in the final microscope slide.
Great care must be taken by histologists when cutting prostate needle cores. Usually, multiple, shallow levels are taken and picked up on 5 separate slides. Slides 1, 3 and 5 are stained with H&E. Slides 2 and 4 are held, unstained, for use in the IHC staining with the PIN 4 cocktail. Preservation of tissue is of the utmost importance.
The resulting stained slides are able to be used to obtain the maximum amount of information regarding any pathology present in these tiny specimens. Pathologists are able to assess and grade the prostate cancer, if present. This information is relayed to the clinician to help determine the treatment and prognosis of the patient.

REFERENCES:
- Theory and Practice of Histological Techniques. JD Bancroft, A Stevens ed. Churchill Livingstone, NY. Fourth edition. 1996
- Theory and Practice of Histotechnology. DC Sheehan, BB Hrapchak. CV Mosby Company, St. Louis. First edition. 1980.
- www.mayoclinic.org


